MDEpiNet Structure

Strategic Areas of Focus
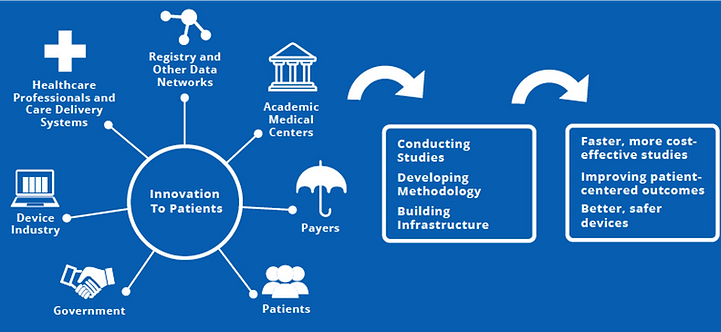
Coordinated Registry Networks (CRNs) are a key MDEpiNet strategy to bring together real-world data from a variety of sources to address the needs of device evaluation for multiple stakeholders. Since 2017, MDEpiNet has been developing learning communities (LC) for CRNs to speed the development and maturity of the networks. LC's main goal is to promote CRN development as a robust source of evidence for device evaluation in multiple areas. The center of the diagram above depicts functions of the MDEpiNet coordinating center. The coordinating center collaborates with various MDEpiNet programs that serve as laboratories for methods development and often leverages the methods and tools developed by them for advancing and maturing CRNs. The MDEpiNet has other programs that are not currently CRNs, but are research and surveillance demonstration initiatives that have a potential to become CRNs.
Key Capabilities
-
Established the collaborative learning communities for national and international CRNs to address the limitations of traditional registries and data repositories by building linked data systems from multiple sources
-
Major programs that focus on patient-led roundtable, unique device identification adoption, data interoperability, novel methodology development and application, international registry consortia collaborative studies
-
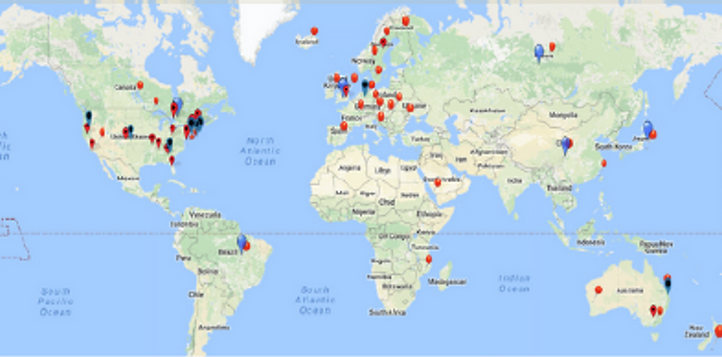
Over 160 partnering public and private sector organizations from the health technology ecosystem globally (academia, professional associations, registries, industry, patient organizations, health systems and more)
-
Over 120 national/regional registries from 45 countries
-
12 National CRNs and 4 International Registry Consortia
-
Over 780 clinical experts across clinical areas
-
Over 250 methodologists (statisticians, epidemiologists, data scientists, informaticians, health care researchers and more)
-
Access to hundreds of millions of patients (Registries, National and State claims data, National and International electronic health records and Integrated Health Care Delivery Systems)
-
MDEpiNet HIVE (High Performance Integrated Virtual Environment), with blockchain solutions with analytical tools for cloud computing
-
International chapters in Europe, Asia and Australia
-
Over 250 core publications in peer review journals featuring methods, infrastructure and partnerships in real world evidence space (over the past 6 years)
-
Recognized as the National Evaluation System for health Technology Coordinating Center (NESTcc) data network collaborator
More Details
Example CRN
MDEpiNet Vascular CRN – VISION which encompasses data sets from national registry (Vascular Quality Initiative), claims data, NY State data (SPARCS), PCORNet, Mobile Apps and clinical trial data tailored for multiple uses.

International Presence